Our Approach
Our deep understanding of the key biological underpinnings of cancer and ability to identify, interrogate, and successfully drug intracellular enzyme targets enables us to advance unique small molecule therapeutics with expansive potential.
Leveraging our industry-leading expertise in genomic instability, we have built a flexible model that allows for a diversity of approaches with the goal of developing transformative biomarker-driven cancer medicines. Our therapies are designed for high-impact oncogenic targets with the potential to benefit large patient populations.
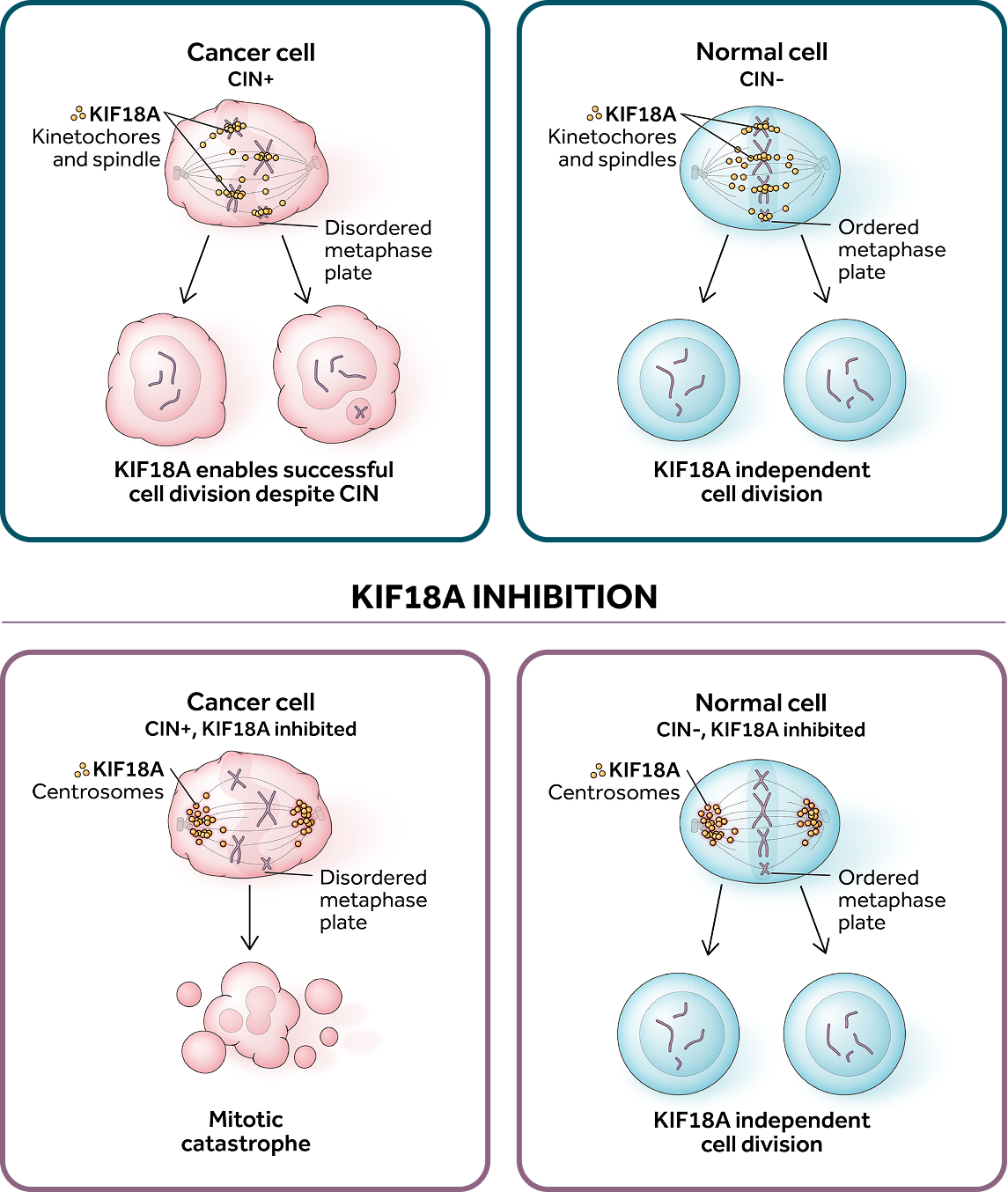
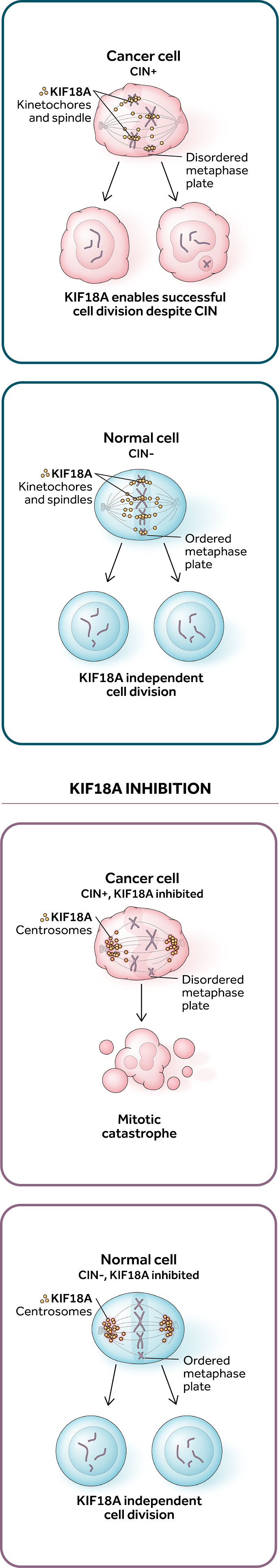
Our lead program, ATX-295, is a potential best-in-class inhibitor for KIF18A, a mitotic kinesin motor protein critical for cell division in select tumors with chromosomal instability, or CIN. KIF18A inhibitor treatment results in rapid cell death for cancers with an abnormal number of chromosomes (aneuploid) in vitro and in vivo, while cells with normal numbers of chromosomes (euploid) are unaffected. ATX-295 may address a large patient population across several cancer indications, including ovarian and squamous non-small cell lung cancer.
KIF18A is a selectively-essential target for tumors
with high levels of chromosomal instability (CIN)